No products in the cart.
NH4Cl Explained: A Comprehensive Guide to Ammonium Chloride’s Properties, Uses, and Safety Precautions
Contents
hide
Ammonium chloride, or NH4Cl, is a versatile chemical compound that plays a crucial role in various industries, from pharmaceuticals to food production. But what exactly is NH4Cl, and how does it work its magic? In this in-depth guide, we’ll delve into the world of NH4Cl, covering its properties, uses, and safety precautions. By the end of this article, you’ll be well-versed in the ins and outs of this fascinating compound and equipped with the knowledge to handle it with confidence.
Whether you’re a scientist, a chef, or a curious enthusiast, this guide has something for everyone. We’ll explore the pH of NH4Cl in water, its potential as a neutralizing agent, and its safety profile. You’ll learn about its common uses, including its role in fertilizers and its applications in cooking. We’ll also examine the chemical formula of NH4Cl, its appearance, and what happens when it’s heated. So, let’s get started on this journey into the world of NH4Cl!
By the end of this article, you’ll be able to:
* Understand the pH of NH4Cl in water and its implications
* Learn whether NH4Cl can be used as a neutralizing agent and its effectiveness
* Discover common uses of NH4Cl and its applications in various industries
* Understand the safety precautions to take when handling NH4Cl
* Explore the chemical formula, appearance, and properties of NH4Cl
* Learn about the potential risks and hazards associated with NH4Cl
So, what are you waiting for? Dive into the world of NH4Cl and uncover its secrets.
🔑 Key Takeaways
- NH4Cl is a strong electrolyte and can be used as a neutralizing agent for acidic spills.
- NH4Cl can be used as a fertilizer, but it should be handled with caution due to its potential toxicity.
- NH4Cl is safe for use in food preparation when used in moderation and handled properly.
- The pH of NH4Cl in water is around 5.5, making it a weak acid.
- NH4Cl can be used in cooking to add flavor and moisture to dishes.
- When heated, NH4Cl releases hydrogen chloride gas, which can be hazardous if not handled properly.
The pH of NH4Cl in Water: A Key to Understanding its Properties
The pH of NH4Cl in water is around 5.5, making it a weak acid. This property is crucial in understanding its behavior and applications. When NH4Cl is dissolved in water, it dissociates into ammonium ions and chloride ions. The ammonium ions are the primary contributors to the acidic nature of NH4Cl. This property makes NH4Cl an effective neutralizing agent for acidic spills.
In a neutralizing agent, the goal is to raise the pH of a solution to a safer level. NH4Cl can achieve this by reacting with the acidic substance and forming a salt. However, it’s essential to note that NH4Cl is not a strong base and should not be used to neutralize highly acidic substances. Instead, it’s more suited for neutralizing weak acids or slightly acidic substances.
For example, if you’re dealing with a spill of a weak acid like vinegar (acetic acid), NH4Cl can be an effective neutralizing agent. Simply mix the NH4Cl with water to create a solution, and then apply it to the spill. The NH4Cl will react with the acetic acid, raising the pH of the solution and making it safer to handle.
NH4Cl as a Neutralizing Agent: Effectiveness and Limitations
As we discussed earlier, NH4Cl can be used as a neutralizing agent for acidic spills. However, its effectiveness depends on the type and concentration of the acidic substance. If the acid is highly concentrated or strong, NH4Cl may not be sufficient to neutralize it. In such cases, stronger bases like sodium hydroxide (NaOH) or calcium hydroxide (Ca(OH)2) may be required.
On the other hand, if the acid is weak or slightly acidic, NH4Cl can be an effective neutralizing agent. For instance, if you’re dealing with a spill of a weak acid like citric acid, NH4Cl can help raise the pH of the solution to a safer level. However, it’s crucial to remember that NH4Cl is not a substitute for proper safety protocols. Always wear protective gear, including gloves and goggles, when handling acidic substances and their neutralizing agents.
To illustrate this point, consider a scenario where you’re working with a weak acid like citric acid in a laboratory setting. If you accidentally spill the acid, you can use NH4Cl to neutralize it. Simply mix the NH4Cl with water to create a solution, and then apply it to the spill. The NH4Cl will react with the citric acid, raising the pH of the solution and making it safer to handle.
Is NH4Cl Harmful to Humans if Ingested?
NH4Cl is generally considered safe for human consumption in small amounts. However, ingesting large quantities can be hazardous. The ammonium ion (NH4+) is the primary contributor to the toxicity of NH4Cl. When ingested, the ammonium ion can cause a range of symptoms, including nausea, vomiting, and diarrhea.
In severe cases, ingesting high amounts of NH4Cl can lead to more serious health issues, such as respiratory failure or even death. It’s essential to handle NH4Cl with caution and keep it out of reach of children and pets.
For example, if you’re working in a laboratory setting and accidentally ingest a small amount of NH4Cl, you may experience mild symptoms like nausea or vomiting. However, if you ingest a large quantity, you may require medical attention. In such cases, it’s crucial to seek immediate help and provide the healthcare provider with information about the ingested substance.
What are Some Common Uses of NH4Cl?
NH4Cl has a wide range of applications across various industries. One of its most common uses is as a fertilizer in agriculture. NH4Cl helps to provide ammonium ions to plants, which are essential for growth and development. However, it’s essential to use NH4Cl in moderation, as excessive application can lead to toxicity.
In addition to its use as a fertilizer, NH4Cl is also used in food production as a leavening agent. It helps to release carbon dioxide gas, which causes bread dough to rise. NH4Cl is also used in the pharmaceutical industry as an excipient, helping to stabilize and preserve medications.
To illustrate this point, consider a scenario where you’re working in a bakery and need to add a leavening agent to your bread dough. You can use NH4Cl to release carbon dioxide gas, causing the dough to rise. Simply mix the NH4Cl with water to create a solution, and then add it to the dough. The NH4Cl will help to create a light and fluffy texture.
Can NH4Cl be Used in Cooking?
Yes, NH4Cl can be used in cooking to add flavor and moisture to dishes. However, it’s essential to use it in moderation and handle it properly. NH4Cl can be used to make various types of food, including soups, stews, and sauces.
For example, you can use NH4Cl to make a flavorful soup by adding it to the broth. Simply mix the NH4Cl with water to create a solution, and then add it to the soup. The NH4Cl will help to add a salty, umami flavor to the soup.
When using NH4Cl in cooking, it’s crucial to follow proper safety protocols. Always wear protective gear, including gloves and goggles, and ensure that you’re working in a well-ventilated area. Additionally, be sure to handle the NH4Cl with care, as it can be hazardous if not handled properly.
Is NH4Cl Safe for Use in Food Preparation?
Yes, NH4Cl is generally considered safe for use in food preparation when used in moderation and handled properly. However, it’s essential to follow proper safety protocols and ensure that you’re working in a clean and well-ventilated area.
When using NH4Cl in food preparation, it’s crucial to consider the potential risks and hazards associated with it. For example, if you’re pregnant or breastfeeding, you should avoid using NH4Cl in food preparation, as it may pose a risk to your health.
To illustrate this point, consider a scenario where you’re preparing a meal for a large group of people. You can use NH4Cl to add flavor and moisture to the dish. However, before using it, make sure to follow proper safety protocols and ensure that you’re working in a clean and well-ventilated area.
The Chemical Formula of NH4Cl: A Key to Understanding its Properties
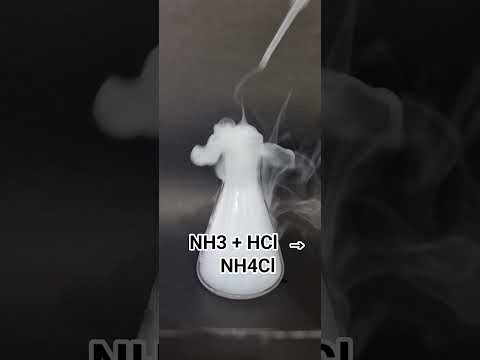
The chemical formula of NH4Cl is NH4Cl, which consists of an ammonium ion (NH4+) and a chloride ion (Cl-). This formula is crucial in understanding the properties and behavior of NH4Cl. The ammonium ion is the primary contributor to the acidic nature of NH4Cl, while the chloride ion helps to stabilize the compound.
In a chemical compound, the formula is a shorthand way of representing the elements present and their proportions. For example, the formula H2O represents water, which consists of two hydrogen atoms and one oxygen atom. Similarly, the formula NH4Cl represents ammonium chloride, which consists of an ammonium ion and a chloride ion.
The Appearance of Solid NH4Cl: A Physical Description
Solid NH4Cl is a white or colorless crystalline substance. It has a characteristic odor, often described as salty or ammoniacal. The appearance of NH4Cl can vary depending on the purity and concentration of the compound.
When NH4Cl is in its pure form, it appears as a white or colorless crystalline solid. However, if it’s contaminated with other substances or has a high concentration of impurities, it may appear as a yellowish or brownish solid. In such cases, it’s essential to handle the NH4Cl with caution and ensure that you’re working in a well-ventilated area.
What Happens When NH4Cl is Heated?
When NH4Cl is heated, it decomposes into ammonium chloride gas and hydrogen chloride gas. This decomposition reaction occurs when the NH4Cl is exposed to high temperatures, often above 200°C.
The decomposition reaction of NH4Cl is an exothermic process, releasing heat and energy. This reaction is crucial in understanding the behavior and properties of NH4Cl. When NH4Cl is heated, it releases hydrogen chloride gas, which can be hazardous if not handled properly.
For example, if you’re working in a laboratory setting and need to heat NH4Cl, you should take necessary precautions to avoid exposure to the hydrogen chloride gas. Wear protective gear, including gloves and goggles, and ensure that you’re working in a well-ventilated area. Additionally, be sure to follow proper safety protocols and ensure that you’re handling the NH4Cl with care.
Is NH4Cl a Strong Electrolyte?
Yes, NH4Cl is a strong electrolyte. An electrolyte is a substance that conducts electricity when dissolved in water. NH4Cl is a strong electrolyte because it dissociates into ions when dissolved in water, allowing it to conduct electricity.
The dissociation of NH4Cl into ions is an essential property of strong electrolytes. When NH4Cl is dissolved in water, it breaks down into an ammonium ion (NH4+) and a chloride ion (Cl-). These ions are then free to move and conduct electricity, making NH4Cl a strong electrolyte.
To illustrate this point, consider a scenario where you’re working in a laboratory setting and need to conduct an experiment involving electrolysis. You can use NH4Cl as a strong electrolyte to facilitate the movement of ions and conduct electricity.
Can NH4Cl be Used as a Fertilizer?
Yes, NH4Cl can be used as a fertilizer in agriculture. NH4Cl helps to provide ammonium ions to plants, which are essential for growth and development. However, it’s essential to use NH4Cl in moderation, as excessive application can lead to toxicity.
When using NH4Cl as a fertilizer, it’s crucial to follow proper safety protocols and ensure that you’re working in a well-ventilated area. Additionally, be sure to handle the NH4Cl with care, as it can be hazardous if not handled properly.
For example, if you’re working in a greenhouse setting and need to fertilize your plants, you can use NH4Cl to provide them with ammonium ions. Simply mix the NH4Cl with water to create a solution, and then apply it to the plants. The NH4Cl will help to promote healthy growth and development.
What are Some Safety Precautions to Take When Handling NH4Cl?
When handling NH4Cl, it’s essential to take necessary precautions to avoid exposure and injury. Here are some safety precautions to take:
* Wear protective gear, including gloves and goggles, when handling NH4Cl
* Ensure that you’re working in a well-ventilated area to avoid inhaling hydrogen chloride gas
* Follow proper safety protocols and ensure that you’re handling the NH4Cl with care
* Avoid ingesting NH4Cl, as it can be hazardous if consumed in large quantities
* Keep NH4Cl out of reach of children and pets
* Follow proper disposal procedures for NH4Cl to avoid environmental contamination
To illustrate this point, consider a scenario where you’re working in a laboratory setting and need to handle NH4Cl. You should wear protective gear, including gloves and goggles, and ensure that you’re working in a well-ventilated area. Additionally, be sure to follow proper safety protocols and handle the NH4Cl with care.
The pH of NH4Cl in Water: Implications and Applications
As we discussed earlier, the pH of NH4Cl in water is around 5.5, making it a weak acid. This property is crucial in understanding its behavior and applications. When NH4Cl is dissolved in water, it dissociates into an ammonium ion (NH4+) and a chloride ion (Cl-). The ammonium ion is the primary contributor to the acidic nature of NH4Cl.
The pH of NH4Cl in water has significant implications for its applications. For example, if you’re working in a laboratory setting and need to neutralize an acidic substance, NH4Cl can be an effective agent. Simply mix the NH4Cl with water to create a solution, and then apply it to the substance. The NH4Cl will react with the acidic substance, raising the pH of the solution and making it safer to handle.
NH4Cl in Food Production: A Leavening Agent
NH4Cl is commonly used in food production as a leavening agent. It helps to release carbon dioxide gas, which causes bread dough to rise. When NH4Cl is mixed with water, it releases hydrogen ions, which react with the carbon dioxide gas to form carbonic acid. The carbonic acid then reacts with the baking soda (sodium bicarbonate) to release more carbon dioxide gas, causing the dough to rise.
To illustrate this point, consider a scenario where you’re working in a bakery and need to add a leavening agent to your bread dough. You can use NH4Cl to release carbon dioxide gas and cause the dough to rise. Simply mix the NH4Cl with water to create a solution, and then add it to the dough. The NH4Cl will help to create a light and fluffy texture.
The Chemical Properties of NH4Cl: A Key to Understanding its Behavior
NH4Cl is a chemical compound that consists of an ammonium ion (NH4+) and a chloride ion (Cl-). This formula is crucial in understanding the properties and behavior of NH4Cl. The ammonium ion is the primary contributor to the acidic nature of NH4Cl, while the chloride ion helps to stabilize the compound.
The chemical properties of NH4Cl are essential in understanding its behavior and applications. For example, if you’re working in a laboratory setting and need to conduct an experiment involving electrolysis, NH4Cl can be used as a strong electrolyte to facilitate the movement of ions and conduct electricity.
NH4Cl in Pharmaceutical Applications: An Excipient
NH4Cl is commonly used in pharmaceutical applications as an excipient. An excipient is a substance that helps to stabilize and preserve medications. NH4Cl is used in various forms, including tablets, capsules, and solutions.
To illustrate this point, consider a scenario where you’re working in a pharmaceutical laboratory and need to develop a new medication. You can use NH4Cl as an excipient to help stabilize and preserve the medication. Simply mix the NH4Cl with water to create a solution, and then add it to the medication. The NH4Cl will help to create a stable and effective medication.
The Environmental Impact of NH4Cl: A Concern
The environmental impact of NH4Cl is a concern due to its potential to contaminate soil, water, and air. NH4Cl can be released into the environment through various means, including industrial waste, agricultural runoff, and household waste.
To mitigate the environmental impact of NH4Cl, it’s essential to follow proper disposal procedures and ensure that you’re handling the compound with care. Wear protective gear, including gloves and goggles, and ensure that you’re working in a well-ventilated area. Additionally, be sure to follow proper safety protocols and ensure that you’re handling the NH4Cl with care.
NH4Cl in Agriculture: A Fertilizer
NH4Cl is commonly used in agriculture as a fertilizer. NH4Cl helps to provide ammonium ions to plants, which are essential for growth and development. However, it’s essential to use NH4Cl in moderation, as excessive application can lead to toxicity.
To illustrate this point, consider a scenario where you’re working in a greenhouse setting and need to fertilize your plants. You can use NH4Cl to provide them with ammonium ions. Simply mix the NH4Cl with water to create a solution, and then apply it to the plants. The NH4Cl will help to promote healthy growth and development.
❓ Frequently Asked Questions
What is the shelf life of NH4Cl?
The shelf life of NH4Cl depends on various factors, including storage conditions and handling. Generally, NH4Cl can last for several years when stored in a cool, dry place. However, it’s essential to follow proper storage procedures and ensure that you’re handling the compound with care.
If you’re working in a laboratory setting, it’s recommended to store NH4Cl in a sealed container and keep it away from direct sunlight and moisture. Additionally, be sure to follow proper safety protocols and ensure that you’re handling the NH4Cl with care.
In a home setting, it’s essential to store NH4Cl in a cool, dry place and keep it away from children and pets. Avoid exposing NH4Cl to direct sunlight and moisture, as it can cause it to degrade. If you’re unsure about the shelf life of NH4Cl, consult the manufacturer’s instructions or contact a professional for guidance.
Can NH4Cl be used as a de-icer for roads and sidewalks?
Yes, NH4Cl can be used as a de-icer for roads and sidewalks. NH4Cl is effective in lowering the freezing point of water, making it an excellent de-icer. However, it’s essential to follow proper safety protocols and ensure that you’re handling the compound with care.
When using NH4Cl as a de-icer, it’s crucial to follow the manufacturer’s instructions and ensure that you’re working in a well-ventilated area. Wear protective gear, including gloves and goggles, and avoid inhaling the hydrogen chloride gas released by NH4Cl.
Additionally, be sure to follow proper disposal procedures for NH4Cl to avoid environmental contamination. If you’re unsure about the use of NH4Cl as a de-icer, consult the manufacturer’s instructions or contact a professional for guidance.
Can NH4Cl be used as a fire extinguisher?
No, NH4Cl should not be used as a fire extinguisher. NH4Cl is not effective in putting out fires, and it can even exacerbate the situation. Instead, use a fire extinguisher rated for the type of fire you’re dealing with.
When dealing with fires, it’s essential to follow proper safety protocols and ensure that you’re handling the situation with care. Wear protective gear, including gloves and goggles, and avoid inhaling smoke or other hazardous substances. If you’re unsure about how to handle a fire, consult the manufacturer’s instructions or contact a professional for guidance.
Can NH4Cl be used as a cleaning agent?
Yes, NH4Cl can be used as a cleaning agent. NH4Cl is effective in removing dirt and grime from surfaces. However, it’s essential to follow proper safety protocols and ensure that you’re handling the compound with care.
When using NH4Cl as a cleaning agent, it’s crucial to follow the manufacturer’s instructions and ensure that you’re working in a well-ventilated area. Wear protective gear, including gloves and goggles, and avoid inhaling the hydrogen chloride gas released by NH4Cl.
Additionally, be sure to follow proper disposal procedures for NH4Cl to avoid environmental contamination. If you’re unsure about the use of NH4Cl as a cleaning agent, consult the manufacturer’s instructions or contact a professional for guidance.
Can NH4Cl be used as a pesticide?
No, NH4Cl should not be used as a pesticide. NH4Cl is not effective in controlling pests, and it can even harm the environment. Instead, use a pesticide rated for the type of pest you’re dealing with.
When dealing with pests, it’s essential to follow proper safety protocols and ensure that you’re handling the situation with care. Wear protective gear, including gloves and goggles, and avoid inhaling pesticides or other hazardous substances. If you’re unsure about how to handle pests, consult the manufacturer’s instructions or contact a professional for guidance.