No products in the cart.
The Ultimate Guide to Lithium Bromide: Properties, Uses, and Safety Precautions
Contents
hide
Lithium bromide is a chemical compound that has been widely used in various industries due to its unique properties. From its high solubility in water to its effectiveness as a flame retardant, lithium bromide has become an essential component in many applications. However, handling this chemical requires careful consideration of its potential risks and hazards. In this comprehensive guide, we will delve into the world of lithium bromide, exploring its properties, uses, and safety precautions. We will also discuss its appearance, manufacturing process, and environmental concerns, providing you with a thorough understanding of this versatile chemical.
Lithium bromide is a white, crystalline powder with a molecular formula of LiBr. It is highly soluble in water, making it an ideal component in various industrial processes. The molar mass of lithium bromide is approximately 86.85 g/mol, which is relatively low compared to other bromide compounds. This low molar mass contributes to its high solubility and reactivity, making it a valuable chemical in many applications.
Whether you are a chemist, an engineer, or simply someone interested in learning more about lithium bromide, this guide is designed to provide you with a detailed and informative overview of this fascinating chemical. We will cover its uses, safety precautions, and environmental concerns, as well as its physical properties and manufacturing process. By the end of this guide, you will have a comprehensive understanding of lithium bromide and its role in various industries.
🔑 Key Takeaways
- Lithium bromide has a molar mass of approximately 86.85 g/mol and is highly soluble in water
- It is used in various industries, including air conditioning, refrigeration, and pharmaceuticals
- Lithium bromide requires careful handling due to its potential risks and hazards
- It is an effective flame retardant and can be used in fire protection systems
- The manufacturing process of lithium bromide involves the reaction of lithium carbonate with hydrobromic acid
- Lithium bromide has environmental concerns due to its potential toxicity and reactivity
- It is essential to follow proper safety precautions when handling lithium bromide, including wearing protective gear and working in a well-ventilated area
Properties and Appearance
Lithium bromide is a white, crystalline powder with a molecular formula of LiBr. It has a melting point of approximately 548°C and a boiling point of 1265°C. Its high solubility in water makes it an ideal component in various industrial processes, including air conditioning and refrigeration systems. The appearance of lithium bromide can vary depending on its purity and crystal structure, but it is generally a white or off-white powder.
In terms of its physical properties, lithium bromide has a density of approximately 3.46 g/cm³ and a refractive index of 1.78. It is also highly hygroscopic, meaning it can absorb moisture from the air, which can affect its purity and reactivity. Understanding the physical properties of lithium bromide is essential for handling and storing it safely and effectively.
Uses and Applications
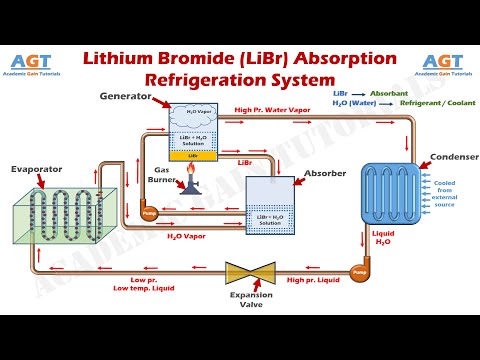
Lithium bromide has a wide range of uses and applications due to its unique properties. One of its primary uses is in air conditioning and refrigeration systems, where it is used as a absorbent to control humidity and temperature. It is also used in pharmaceuticals, where it is used as an intermediate in the production of certain medications.
In addition to its industrial uses, lithium bromide is also used in fire protection systems due to its effectiveness as a flame retardant. It can be used to extinguish fires in electrical equipment, such as computers and televisions, and can also be used in fire suppression systems for buildings and vehicles. The use of lithium bromide in fire protection systems is due to its ability to release bromine gas, which can help to smother fires and prevent them from spreading.
Manufacturing Process
The manufacturing process of lithium bromide involves the reaction of lithium carbonate with hydrobromic acid. This reaction produces lithium bromide and carbon dioxide, which can be separated and purified through various methods. The resulting lithium bromide can be further purified through recrystallization or other methods to produce a high-purity product.
The manufacturing process of lithium bromide requires careful control of temperature, pressure, and reaction conditions to ensure the production of high-quality lithium bromide. The reaction is typically carried out in a large reactor vessel, where the lithium carbonate and hydrobromic acid are mixed and heated to produce the desired reaction. The resulting lithium bromide can then be separated and purified through various methods, including filtration, centrifugation, and drying.
Safety Precautions and Handling
Handling lithium bromide requires careful consideration of its potential risks and hazards. It is essential to wear protective gear, including gloves, goggles, and a face mask, when handling lithium bromide to prevent skin and eye irritation. It is also important to work in a well-ventilated area to prevent inhalation of the dust or fumes.
In addition to personal protective equipment, it is also essential to follow proper handling and storage procedures to prevent accidents and spills. Lithium bromide should be stored in a cool, dry place, away from incompatible materials, such as strong acids and bases. It is also important to follow proper disposal procedures to prevent environmental contamination and harm to humans and animals.
Environmental Concerns
Lithium bromide has environmental concerns due to its potential toxicity and reactivity. It can contaminate soil, water, and air if not handled and disposed of properly. The production and use of lithium bromide can also result in the release of bromine gas, which can contribute to air pollution and harm to humans and animals.
To minimize the environmental concerns associated with lithium bromide, it is essential to follow proper handling, storage, and disposal procedures. This includes using personal protective equipment, working in a well-ventilated area, and following proper disposal procedures to prevent environmental contamination. It is also important to consider alternative chemicals or methods that may be less harmful to the environment.
Transportation and Storage
The transportation and storage of lithium bromide require careful consideration of its potential risks and hazards. It is essential to follow proper packaging and labeling procedures to prevent accidents and spills during transportation. Lithium bromide should be stored in a cool, dry place, away from incompatible materials, such as strong acids and bases.
In addition to proper packaging and labeling, it is also important to follow proper transportation procedures to prevent accidents and spills. This includes using approved containers and vehicles, following proper loading and unloading procedures, and ensuring that the lithium bromide is properly secured during transportation. It is also essential to follow proper storage procedures to prevent environmental contamination and harm to humans and animals.
❓ Frequently Asked Questions
What are the potential health effects of exposure to lithium bromide?
Exposure to lithium bromide can cause skin and eye irritation, as well as respiratory problems. Prolonged exposure can also lead to more serious health effects, including kidney and liver damage. It is essential to follow proper handling and safety precautions to minimize the risk of exposure.
In addition to skin and eye irritation, exposure to lithium bromide can also cause respiratory problems, such as coughing and shortness of breath. This is due to the release of bromine gas, which can irritate the lungs and airways. It is essential to work in a well-ventilated area and follow proper handling and safety precautions to minimize the risk of exposure.
Can lithium bromide be used as a medication?
Lithium bromide is not typically used as a medication, although it can be used as an intermediate in the production of certain medications. Lithium salts, such as lithium carbonate, are commonly used to treat mental health conditions, such as bipolar disorder. However, lithium bromide is not approved for use as a medication and should not be taken without proper medical supervision.
In some cases, lithium bromide may be used in medical research or as a component in certain medical devices. However, its use in these applications is highly regulated and requires proper approval and supervision. It is essential to follow proper handling and safety precautions when working with lithium bromide, even in medical applications.
Is lithium bromide used in food production?
Lithium bromide is not typically used in food production, although it may be used in certain industrial processes, such as water treatment or air conditioning. However, its use in these applications is highly regulated and requires proper approval and supervision.
In some cases, lithium bromide may be used as a component in certain food packaging materials or as a intermediate in the production of certain food additives. However, its use in these applications is highly regulated and requires proper approval and supervision. It is essential to follow proper handling and safety precautions when working with lithium bromide, even in food-related applications.
What are the potential environmental impacts of lithium bromide?
The potential environmental impacts of lithium bromide include contamination of soil, water, and air. The production and use of lithium bromide can result in the release of bromine gas, which can contribute to air pollution and harm to humans and animals.
In addition to air pollution, the production and use of lithium bromide can also result in the contamination of soil and water. This can occur through improper disposal or spills, which can lead to the release of lithium bromide into the environment. It is essential to follow proper handling, storage, and disposal procedures to minimize the risk of environmental contamination.
Can lithium bromide be used as a flame retardant in consumer products?
Lithium bromide can be used as a flame retardant in certain consumer products, such as electronics or textiles. However, its use in these applications is highly regulated and requires proper approval and supervision.
In some cases, lithium bromide may be used as a component in certain flame retardant materials or as a intermediate in the production of certain flame retardant chemicals. However, its use in these applications is highly regulated and requires proper approval and supervision. It is essential to follow proper handling and safety precautions when working with lithium bromide, even in consumer product applications.
What are the potential risks and hazards associated with the transportation of lithium bromide?
The potential risks and hazards associated with the transportation of lithium bromide include accidents and spills, which can result in environmental contamination and harm to humans and animals. It is essential to follow proper packaging and labeling procedures to prevent accidents and spills during transportation.
In addition to accidents and spills, the transportation of lithium bromide can also result in exposure to bromine gas, which can irritate the lungs and airways. It is essential to follow proper handling and safety precautions, including the use of personal protective equipment and proper ventilation, to minimize the risk of exposure.