No products in the cart.
The Atomic Composition of Salt: Unlocking the Secrets of Sodium Chloride
Contents
hide
Salt, or sodium chloride (NaCl), is one of the most ubiquitous substances on Earth. We sprinkle it on our food, use it to preserve meats, and even harvest it from ancient sea beds. But have you ever stopped to think about the tiny building blocks that make up this everyday essential? In this comprehensive guide, we’ll delve into the fascinating world of salt’s atomic composition and explore how it affects its taste, properties, and uses. By the end of this journey, you’ll have a deeper understanding of the science behind salt and its many applications.
🔑 Key Takeaways
- Salt is composed of two atoms: one sodium (Na) and one chlorine (Cl) atom, forming a single molecule of NaCl.
- The atomic composition of salt is crucial to its taste, texture, and chemical properties.
- The sodium and chlorine atoms in salt have distinct roles in its properties and applications.
- The atomic composition of salt can affect its potential health implications and industrial uses.
- Understanding the atomic composition of salt can help you appreciate its unique characteristics and uses more deeply.
The Formation of Salt: A Chemical Reaction
Salt is formed through a process called evaporation, where water containing dissolved minerals is heated, causing the water to evaporate and leave behind a residue of salt. This process can occur naturally, such as in the formation of sea salt, or artificially, like in the production of table salt. The reaction that forms salt is a simple one: the combination of sodium ions (Na+) and chlorine ions (Cl-) results in the formation of a sodium chloride molecule (NaCl). This process is crucial to understanding the atomic composition of salt, as it highlights the fundamental role of sodium and chlorine in its structure.
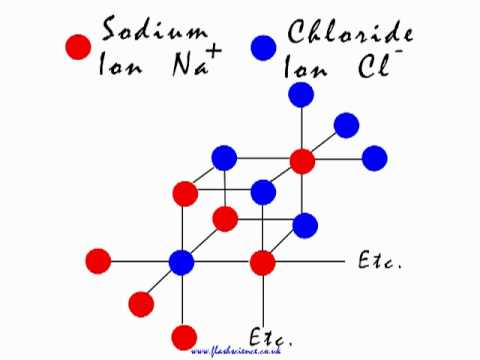
The Atomic Makeup of Salt: A Single Molecule
A single molecule of salt is composed of two atoms: one sodium (Na) and one chlorine (Cl) atom. This may seem simplistic, but the significance of this atomic makeup cannot be overstated. The sodium atom, with its single valence electron, is highly reactive and seeks to share its electron with other atoms. The chlorine atom, with its seven valence electrons, is also highly reactive and seeks to gain one electron to complete its outer shell. The combination of these two atoms results in a stable molecule that is essential to life as we know it. The atomic composition of salt is what gives it its unique properties and applications, from its use as a seasoning to its role in preserving meats.
Significance of the Atomic Makeup of Salt
The atomic makeup of salt is crucial to its taste, texture, and chemical properties. The sodium and chlorine atoms in salt work together to create a unique flavor profile that is both salty and slightly bitter. The texture of salt is also influenced by its atomic composition, as the sodium and chlorine atoms create a crystal structure that is both hard and brittle. Additionally, the atomic makeup of salt affects its chemical properties, such as its solubility and reactivity. Understanding the significance of the atomic makeup of salt can help you appreciate its unique characteristics and uses more deeply.
How Atoms in Salt Contribute to Its Taste
The atoms in salt contribute to its taste through a process called ionization. When salt is dissolved in water, the sodium and chlorine atoms break apart, releasing ions that interact with the taste buds on our tongues. The sodium ions activate the taste receptors on our tongues, sending a signal to the brain that we experience as saltiness. The chlorine ions, on the other hand, contribute to a slightly bitter or sour taste. The combination of these ions creates a unique flavor profile that is both salty and slightly bitter.
Health Implications Related to the Atoms in Salt
The atoms in salt can have potential health implications, particularly when consumed in excess. The high sodium content in salt can lead to high blood pressure, heart disease, and stroke. Additionally, the chlorine content in salt can contribute to digestive issues, such as bloating and gas. However, it’s worth noting that the health implications of salt’s atoms are largely related to its use as a seasoning, rather than its atomic composition itself.
Can the Number of Atoms in Salt Affect Its Chemical Properties?
Yes, the number of atoms in salt can affect its chemical properties. The atomic composition of salt is crucial to its solubility, reactivity, and other chemical properties. However, it’s worth noting that the number of atoms in salt is fixed at two sodium and one chlorine atom per molecule. This is due to the chemical bond between the sodium and chlorine atoms, which creates a stable molecule that is essential to life as we know it.
Interesting Facts About the Atoms in Salt
The atoms in salt have several interesting properties. For example, the sodium atom is highly reactive and seeks to share its electron with other atoms. The chlorine atom, on the other hand, is highly reactive and seeks to gain one electron to complete its outer shell. This chemical reactivity is what makes salt so essential to life, as it plays a crucial role in many biological processes. Additionally, the atomic composition of salt is responsible for its unique properties, such as its taste, texture, and chemical properties.
How Salt Compares to Other Compounds in Terms of Its Atomic Composition
Salt is unique in its atomic composition, as it is composed of two atoms: one sodium and one chlorine atom. This is in contrast to other compounds, such as water, which is composed of two hydrogen atoms and one oxygen atom. The atomic composition of salt is what gives it its unique properties and applications, from its use as a seasoning to its role in preserving meats.
Industrial Applications Related to the Atoms in Salt
The atoms in salt have several industrial applications, particularly in the production of chemicals and pharmaceuticals. The sodium and chlorine atoms in salt are used as reactants in the production of a wide range of chemicals, including bleach, paper, and textiles. Additionally, the atomic composition of salt is used in the production of pharmaceuticals, such as antacids and laxatives.
Can the Atomic Composition of Salt Be Manipulated for Specific Purposes?
Yes, the atomic composition of salt can be manipulated for specific purposes. For example, the addition of other elements, such as iodine or potassium, can create new compounds with unique properties. Additionally, the use of different manufacturing processes can affect the atomic composition of salt, resulting in different properties and applications.
How Atoms in Salt Contribute to Its Physical Appearance and Texture
The atoms in salt contribute to its physical appearance and texture through a process called crystallization. When salt is cooled, the sodium and chlorine atoms come together to form a crystal structure that is both hard and brittle. The crystal structure of salt is responsible for its unique texture and appearance, which is both visually appealing and tactilely engaging.
❓ Frequently Asked Questions
What is the difference between sea salt and table salt?
Sea salt and table salt have different atomic compositions due to their production processes. Sea salt is produced through the evaporation of seawater, resulting in a product that is closer to its natural state. Table salt, on the other hand, is produced through the refining of rock salt, resulting in a product that is highly processed and purified. While the atomic composition of both products is similar, the processing differences can affect their properties and uses.
Can I use salt as a preservative in my cooking?
Yes, salt can be used as a preservative in cooking, particularly for meats and vegetables. The sodium content in salt helps to inhibit the growth of bacteria and other microorganisms, extending the shelf life of your food. However, it’s worth noting that the use of salt as a preservative should be done in moderation, as excessive salt consumption can have negative health implications.
How can I reduce the sodium content in my salt?
There are several ways to reduce the sodium content in your salt, including using kosher salt or sea salt, which have lower sodium content than table salt. You can also try using other seasonings, such as herbs and spices, to add flavor to your food without the need for salt. Additionally, you can try using salt substitutes, such as potassium chloride, which have similar flavor profiles to salt but with lower sodium content.
What is the role of salt in our bodies?
Salt plays a crucial role in our bodies, particularly in maintaining fluid balance and regulating blood pressure. The sodium content in salt helps to balance the amount of water in our bodies, while the chloride content helps to regulate the pH level of our blood. Additionally, salt is essential for the proper functioning of our nervous system and muscles.
Can I use salt as a face mask?
Yes, salt can be used as a face mask, particularly for its exfoliating and antibacterial properties. The sodium content in salt helps to break down dead skin cells, while the chloride content helps to regulate the pH level of the skin. To use salt as a face mask, mix 1 tablespoon of salt with 2 tablespoons of water and apply it to your face for 5-10 minutes before rinsing with warm water.
What is the difference between salt and sugar?
Salt and sugar have different atomic compositions due to their chemical structures. Salt is composed of sodium and chloride atoms, while sugar is composed of carbon, hydrogen, and oxygen atoms. The chemical properties and uses of salt and sugar are also distinct, with salt being used as a seasoning and preservative, while sugar is used as a sweetener and energy source.